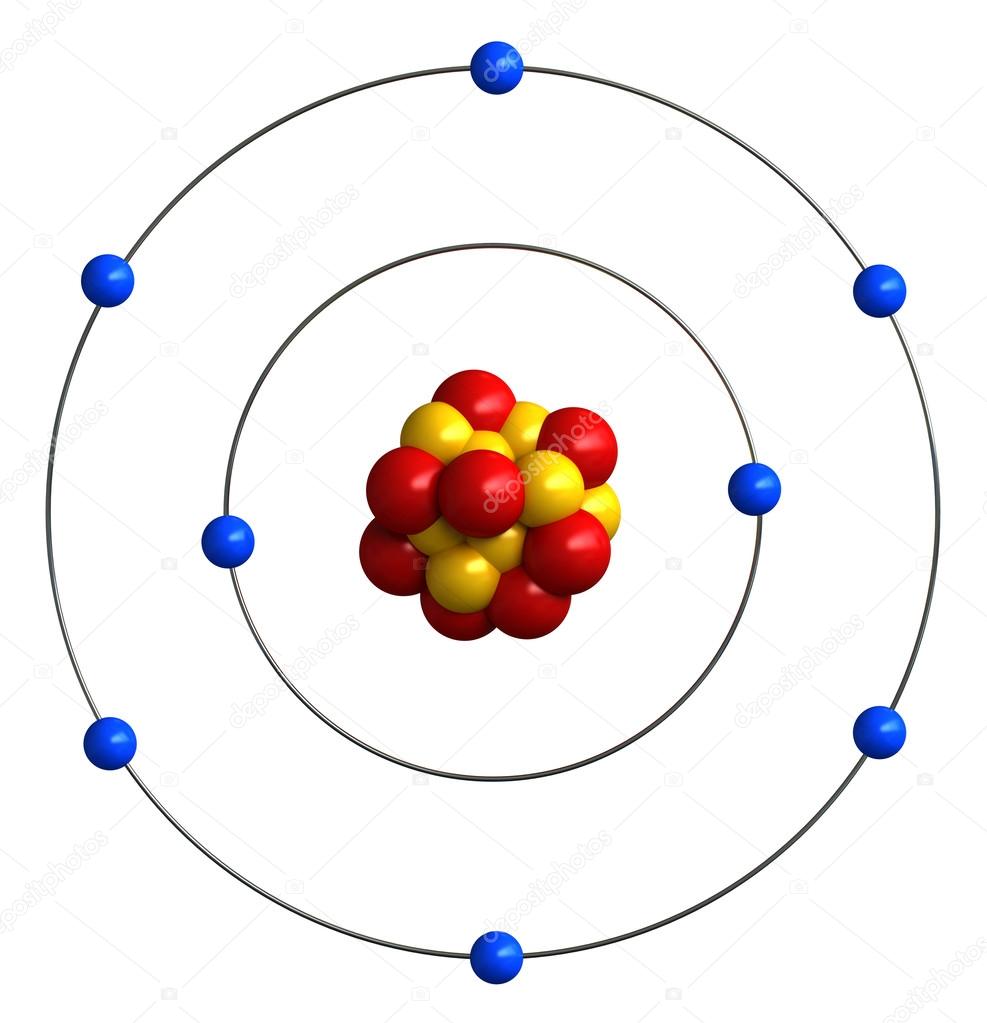
It comes to rest in the \(n = 6\) orbit, so \(n_2 = 6\). (b) The energy of the orbit becomes increasingly less negative with increasing n. 2: The Bohr Model of the Hydrogen Atom (a) The distance of the orbit from the nucleus increases with increasing n. Back in the early days of quantum mechanics, Bohr constructed a model of the hydrogen atom that worked surprisingly well considering its simplicity. Use a paintbrush to make 16 dots on the nucleus, eight. In this state the radius of the orbit is also infinite. In this video well look at the atomic structure and Bohr model for the Oxygen atom (O). This page titled 5.1: The Bohr Model is shared under a CC BY-SA 4.0 license and was authored, remixed, and/or curated by Tom Weideman directly on the LibreTexts platform. Push the nucleus label into the top of the center sphere. Push the oxygen atom label into the top of the largest sphere. In atomic physics, the Bohr model or RutherfordBohr model of the atom, presented by Niels Bohr and Ernest Rutherford in 1913, consists of a small, dense nucleus surrounded by orbiting electrons. Push the electron label into one of the smallest sphere-halves orbiting the oxygen nucleus. In this case, the electron starts out with \(n = 4\), so \(n_1 = 4\). Roll the edge of each strip around a toothpick and secure with glue or tape. What is the energy (in joules) and the wavelength (in meters) of the line in the spectrum of hydrogen that represents the movement of an electron from Bohr orbit with n = 4 to the orbit with n = 6? In what part of the electromagnetic spectrum do we find this radiation? Note that Bohr stated that electrons in the atom follow elliptical orbits (not. The Bohr model, named after scientist Neils Bohr, is a way of illustrating the structure of the atom and the location of its subatomic particles. It has subatomic particles within it called protons, neutrons, and electrons. \): Calculating Electron Transitions in a One–electron System In 1913, Danish physicist Niels Bohr applied Max Planck’s quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ( Figure 1 ). A Model for Atomic Structure: An atom is the smallest particle that makes up matter.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
